purpose
To find plant materials that contain active ingredients that will inhibit the growth of bacteria.
materials
- Balance, weigh boat, lab scoops
- LB broth base
- Media bottles, 250 mL
- Sterilizer/autoclave
- Water bath, 37*C, shaking
- Sterile LB agar
- Laminar flow hood and disinfectant
- Plastic safety glasses
- Bunsen burner and gas lighter
- Inoculating loop, Ni/Cr wire
- Petri dishes, 60x15mm, sterile
- E. coli JM109 (stock plate)
- Plant specimen
- Mortar and pestle
- Pipet, 10 mL and pump
- Plastic funnels, short-stemmed
- Filter paper disks, 5mm diameter
- 100 mL beakers
- Syringe, 10 mL and filter, 0.2 micrometers
- Reaction tubes and rack, 1.7 mL
- Methanol, absolute
- Pipet, 1 mL and pump
- Dry block heater/heat block
- Forceps, fine-tipped
- Ampicillin
- Glass spreader
- Incubator oven, 37*C
Procedure
Part II
4. Grind 2 g of plant tissue (leaves) with 10 mL of deionized water in mortar and pestle, and let sit for 3 minutes. Filter in 11 cm funnel, sterilize extract with syringe filter, and collect 1 mL of extract in labelled 1.7 mL microtube.
5. Repeat Step 4, except replace deionized water with methanol. Place 1.7 mL tube with 1 mL of methanol extract in 65*C heat block (caps open) for 24 hours to evaporate methanol. Reconstitute dry matter in microtube with 1 mL of deionized water.
6. Repeat Step 4 and 5 for six samples and label them.
7. Drop filter paper disks in each filtered extract tube using sterile forceps (sterilized by being flamed in alcohol).
8. Prepare three negative control disks of only methanol and sterile and distilled water.
9. Prepare six positive control disks of ampicillin solution.
10. Allow disks to be saturated with the extract (perhaps overnight).
11. Close tubes. Store all samples at 4*C until ready to use.
Part III
12. Transfer 1 mL of the E. Coli broth to middle of Petri dish with sterile pipet. Sterilize spreading loop with alcohol and flame and spread bacteria culture in Petri dish. Cover and let culture soak in agar for at least 15 minutes.
13. Separating the methanol-extracted samples and the water-extracted samples in different dishes, place one disk on each quadrant at least 2 cm away from edge of dish. Block excess liquid before placing disks in.
14. Repeat step 13 twice. (3 methanol extraction replicates and 3 deionized water extraction replicates)
15. Place one of negative control disks in center of the appropriate plate. Place positive control disk with ampicillin in another quadrant of each plate.
16. Finish with 6 Petri plates with a negative control in the center, a positive control, and 3 sample disks. Record which solvents and plant extracts are in each quadrant. Let soak for a few minutes.
17. Ensure disks adhere to surface of agar. Invert the plates and incubate at 37*C for 24 to 48 hours.
18. After incubation, look for at the plates with plant extract disks for zones of inhibition, clear area formed by inhibitory (decrease in action) action of a substance in the plant material around the disk. Photograph the plates, labeling any inhibition of bacterial growth.
19. Create a data table for the replicates and averages. Include descriptions of the bacterial lawn around each disk. Record the diameter and clarity of any cleared areas around the disks in quantitative measurements.
4. Grind 2 g of plant tissue (leaves) with 10 mL of deionized water in mortar and pestle, and let sit for 3 minutes. Filter in 11 cm funnel, sterilize extract with syringe filter, and collect 1 mL of extract in labelled 1.7 mL microtube.
5. Repeat Step 4, except replace deionized water with methanol. Place 1.7 mL tube with 1 mL of methanol extract in 65*C heat block (caps open) for 24 hours to evaporate methanol. Reconstitute dry matter in microtube with 1 mL of deionized water.
6. Repeat Step 4 and 5 for six samples and label them.
7. Drop filter paper disks in each filtered extract tube using sterile forceps (sterilized by being flamed in alcohol).
8. Prepare three negative control disks of only methanol and sterile and distilled water.
9. Prepare six positive control disks of ampicillin solution.
10. Allow disks to be saturated with the extract (perhaps overnight).
11. Close tubes. Store all samples at 4*C until ready to use.
Part III
12. Transfer 1 mL of the E. Coli broth to middle of Petri dish with sterile pipet. Sterilize spreading loop with alcohol and flame and spread bacteria culture in Petri dish. Cover and let culture soak in agar for at least 15 minutes.
13. Separating the methanol-extracted samples and the water-extracted samples in different dishes, place one disk on each quadrant at least 2 cm away from edge of dish. Block excess liquid before placing disks in.
14. Repeat step 13 twice. (3 methanol extraction replicates and 3 deionized water extraction replicates)
15. Place one of negative control disks in center of the appropriate plate. Place positive control disk with ampicillin in another quadrant of each plate.
16. Finish with 6 Petri plates with a negative control in the center, a positive control, and 3 sample disks. Record which solvents and plant extracts are in each quadrant. Let soak for a few minutes.
17. Ensure disks adhere to surface of agar. Invert the plates and incubate at 37*C for 24 to 48 hours.
18. After incubation, look for at the plates with plant extract disks for zones of inhibition, clear area formed by inhibitory (decrease in action) action of a substance in the plant material around the disk. Photograph the plates, labeling any inhibition of bacterial growth.
19. Create a data table for the replicates and averages. Include descriptions of the bacterial lawn around each disk. Record the diameter and clarity of any cleared areas around the disks in quantitative measurements.
results
|
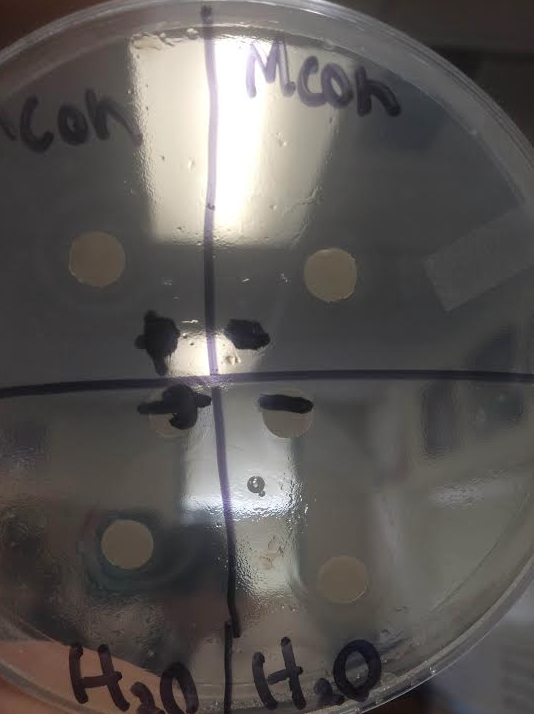
None of the extracts on my plate were successful in inhibiting growth. The MeOH extracts had clear rings around them, but also had bacterial growth next to the filter indicating they were ineffective. The rings around the MeOH samples were about 1mm thick.
The water samples were also ineffective, though the rings around them were about 2mm thick. Their thickness also varied and the rings were not even. Both controls worked successfully; water had bacteria right up next to it and the Ampicillin filter had no bacterial growth around it. If I were to do this experiment again, I would do the same thing but use a different plant from my sate and see if that plant has properties that inhibit microbial growth. My site has many plants and it would be interesting to see which ones do and which ones don't. |
Date Analysis and conclusion
All of the extracts gave me a Negative result. Both of the MeOH extracts and both of the H2O extracts gave me negative answers. My controls did work as expected. The positive control had a clear ring around it. The negative control was a weak negative because the bacteria grew touching the filter but just outside of that was a thin clear ring with no bacteria. I am not sure if any errors might have changed my results, but if i were able to i would conduct the experiment multiple times to see if i got the same results. In the future, I would test more than two filters per extract because there are many factors that could have changed my results.
Thinking Like A Biotechnician Questions
1. If an extract gives a negative result in the antimicrobial assay, the extract is not an antimicrobial assay because the bacteria grows right off of the filter. This means it cannot fight off the bacteria. If it were a medicine, the bacteria would continue to attack.
2. In preparing the sample disks, some of the methanol extractions smell like alcohol. This is a problem because alcohol sterilizes substances, therefore it damages the experiment. The methanol extract would not be the one that is tested if it was sterilized.
3. Each extract may have one or more compounds in it. We can identify the exact compound in an extract that is causing the antimicrobial action by isolating each compound. We could use chromatography to separate each compound in order to test each compound in the extract for antimicrobial action. After isolating each compound, we would do the same experiment above to test for positive and negative results.
Thinking Like A Biotechnician Questions
1. If an extract gives a negative result in the antimicrobial assay, the extract is not an antimicrobial assay because the bacteria grows right off of the filter. This means it cannot fight off the bacteria. If it were a medicine, the bacteria would continue to attack.
2. In preparing the sample disks, some of the methanol extractions smell like alcohol. This is a problem because alcohol sterilizes substances, therefore it damages the experiment. The methanol extract would not be the one that is tested if it was sterilized.
3. Each extract may have one or more compounds in it. We can identify the exact compound in an extract that is causing the antimicrobial action by isolating each compound. We could use chromatography to separate each compound in order to test each compound in the extract for antimicrobial action. After isolating each compound, we would do the same experiment above to test for positive and negative results.